The Internet of Medical Things (IoMT) is one of the most rapid-developing technologies in the healthcare market, and there are just reasons for that. Medical device software development not only improves the quality of care but also makes it easier for the hospitals and doctors to perform their duties and manage their workload. However, like with any fast-growing technologies, it faces the IoMT challenges that, if not seen to, may defy all the positive effects it has on the whole healthcare system. This article is written by Empeek, an experienced company in the development of IoT for healthcare. Let’s take a look together at what IoMT is, what its position in the global market is, what the challenges are in the IoMT sector and how to solve them.
Medical Device Industry and IoMT
The medical device industry is a broader term to denote manufacturing of all devices used in the healthcare sector designed to diagnose and treat patients within global healthcare systems. Medical devices range from simple ones like thermometers and bandages to sophisticated programmable pacemakers, imaging, and scanning systems. Major product medical device product categories include:
- Surgical implants and instruments
- Medical supplies
- Electro-medical equipment
- In-vitro diagnostic equipment and reagents,
- Irradiation apparatuses
- Dental goods.

Where does IoMT stand here?
The Internet of Medical Things (IoMT) means the software and programming solutions and technologies that connect the physical and digital world of healthcare. In other words, IoMT represents the solutions that make possible the transition of data from various medical devices to the system that connects providers with patients and devices above. In this way, a real-time health monitoring system creates a whole new level of cooperation between the involved parties.
IoMT Classification and Market Opportunities
IoT use cases in healthcare can be classified into 3 major categories: by product, by application, and by end-users. Let’s review each.
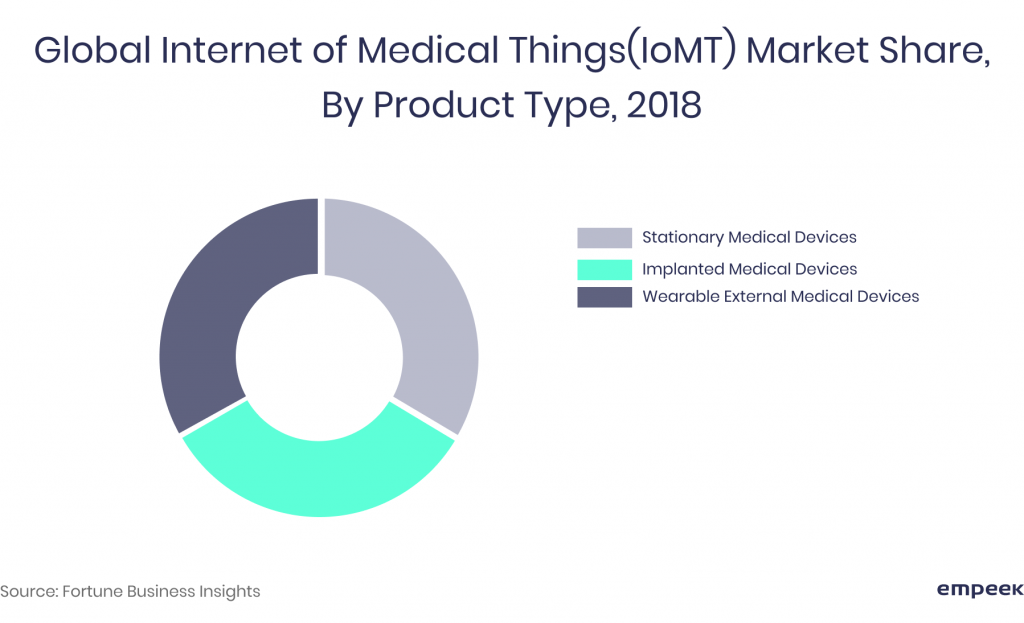
According to the classification by product type, it includes:
- Wearable external devices (skin patches, insulin pumps, blood glucose monitors, etc.)
- Implanted medical devices (pacemakers, implantable cardioverter defibrillators (ICDs), etc.)
- Stationary devices (home monitoring devices, connected imaging devices, scanning machines like MRI devices, CT scanners, X-ray, mammography machines).
Based on application, they are divided into:
- Telemedicine (the highest IoMT market share)
- Medication management
- Patient monitoring
- Other
Finally, the classification based on the end-user:
- Healthcare providers
- Patients
- Government authorities
- Others
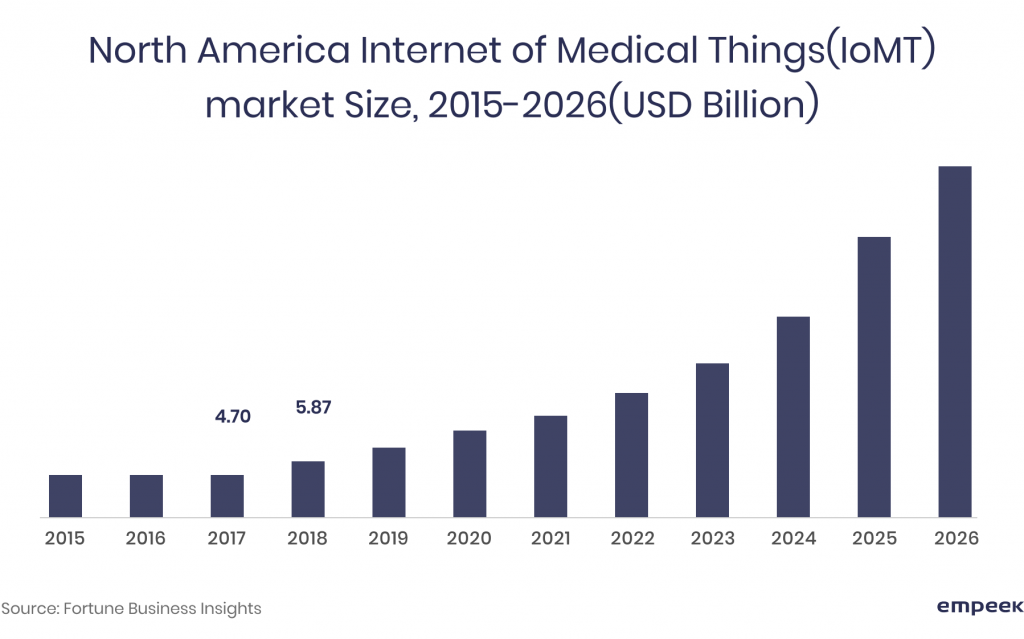
IoMT shows an enormous 28% compound annual growth rate (CAGR) and is expected to reach $142.45 billion by 2026. The reason? Its efficacy in increasing the speed and accuracy of diagnosis and treatments, monitoring and modifying the patient’s behavior and health status in real-time. For organizations, IoMT is valuable for improving their operational productivity by ‘streamlining clinical processes, information, and workflows’. According to Goldman&Sachs, IoMT can potentially save the healthcare industry up to $300 billion annually due to remote monitoring, telemedicine, and improved medication adherence.
This exponential growth and plenty of users involved means not only the benefits of IoMT for the healthcare businesses. The complex infrastructure, the number of involved parties, and the value of the information it deals with, also mean the challenges and possible internet of medical things risks. Below we review the most urgent ones that have to be addressed before IoMT implementation or development.
Challenges in IoMT and How to Overcome Them
IoMT Challenge 1: Cybersecurity
Undoubtedly, connected medical devices cybersecurity is among the most dangerous IoT challenges for medical device industry. Why so? The reason lies in the quality of data received/transmitted via medical devices and technology used. It’s not only the results of the ICD trackers, one’s period calendar, or anything related to the protected health information (PHI) – it also can be the patient’s personal or financial data like DoB, name, social security number, bank account details, etc. The graph below shows the most frequent causes of data breaches in 2020. And it does look like something that the IoT developers should note.
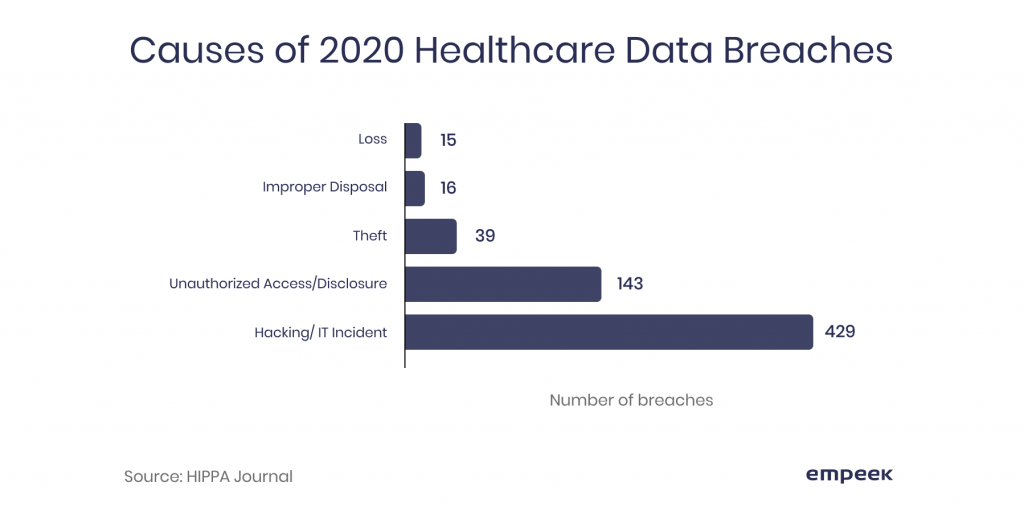
Hacking and IT incidents are credited as the most frequent reasons for unauthorized data exposure. As IoMT deals with the transmission of data, it becomes its vulnerable spot.
Solution: Develop the Product with HIPAA Compliance in Mind
And not because it’s step zero to get to the market and become FDA-approved but because it prevents the patient’s data from becoming an exchange item on the data black market. At Empeek, we pay attention to the HIPAA medical device software requirements regarding the technologies we use for gathering, storing, or transmitting data, as the failure to comply with the technical standards will lead to legal consequences, at the very least.
IoMT Challenge 2: Interoperability
IoMT integration is useful when the collected information can be further sorted out and used to improve patient’s health outcomes. In most cases, the IoMT system lacks interoperability, thus remaining hard to access when needed. While it may be useful for keeping it safe, it complicates the integration of newer technologies and devices into these systems. As a result, it leads to costly transformation or building new networks from scratch, which proves to be costly.
Solution: Open Platforms and Open-Data Standards
The different departments in healthcare have little to no communication, to say nothing of simultaneous use of networks designed by different companies. With secure open platforms based on open data standards, it will become possible to extend the data interoperability between the various care providers, thus tackling interoperability challenges in IoMT.
IoMT Challenge 3: Device Mobility
In IoMT, if the device can only be stationary and located in one spot to be used for treatment, diagnostic, or data handling, it significantly decreases its performance. Another example: an interrupted network connection, which can switch off the device signaling whatsoever, causing potential life-threatening consequences. Hence, making the IoMT devices mobile and functioning is a field to work on.
Solution: Dual Network Use
When the device configuration allows for interchangeable network use, it improves its performing abilities. Yet again, it should be a safe network switch.

IoMT Challenge 4: Licensing & Regulations
Internet of medical things challenges list also features the issues with getting a license to present the product on the market, adopt it into the medical facility, and search for further market opportunities. The reality is that the majority of the devices aimed at collecting, transmitting, or analyzing information are subjects for compliance regulations and licensing approvals. And these processes may take a lot of time.
Solution: Know Them before Developing a Product
If the developer knows the technology standards that are approved for, let’s say, medical monitoring system, it will speed up the development process and increase the chance of getting a license without having to modify the product. Besides, it will save the healthcare providers a lot of trouble during the audits.
IoMT Challenge 5: Improving the Adoption Scale
While the IoMT business is booming and will continue to grow in the future, the question of its scalability and broader adoption is still in the air. For one thing, not all healthcare organizations, clinicians, and patients understand the added value of connected medical devices and how they can drive better patient outcomes or reduce healthcare costs. Another barrier is the lack of governance standards and evidence-based research that IoMT and interconnectivity of medical devices is a cost-effective solution.
Solution: Invest in Research, Improve Awareness, Ensure Cooperation
Ensuring that the devices are intuitive and easy to use, together with providing training to staff to embed the skills needed to optimize the use of the technology, is what every medical establishment can do at any point of IoMT integration. While the governmental issue is more complicated by nature, the companies may show the results of the independent trials, studies, and evidence to increase awareness and do more media exposure.
On the business level, improved collaboration in the form of partnerships and joint ventures will facilitate the effective transmission, aggregation, analysis, and management of data from connected devices. Bonus point? It will improve the stakeholders’ understanding of patient needs and deliver more proactive cost-effective care.

IoMT Challenge 6: Need for Advanced Analytics
Gigabytes of data mean nothing without effective algorithms that sort it out, analyze, and predict possible scenarios. And it’s not only about the tools that help maintain the performance of the medical devices and alert in case anything needs a fix – it’s also about figuring out the possibility of developing heart failure condition or attack. This is where the IoMT can literally save lives.
Solution: Use Machine Learning and AI
IoT in hospitals challenges regarding analytics may be addressed by using advanced algorithms that help to segment & cluster data, recognize patterns and alarm about potential threats. This is the most important principle of Empeek’s approach towards creating any solution, be that medical IoT, EHR, or medical device software.
Final Thoughts
Internet of things challenges in medical device industry can be the stumbling blocks on the way of improving the quality of care, hospital management, patient’s experience, and outcomes. However, addressing them correctly will fully reveal the positive influence that the IoMT holds for healthcare providers, recipients, and other involved parties. Empeek company offers custom healthcare iot solutions development services. One of our recent alike projects was an EMR app to keep families connected. We are ready to join you in this journey and power the devices you use with accurate, reliable, and compliant software solutions. Contact our managers for more information today!